The Adrenal Gland and Stress Responses

Content:
Introduction
The Role of the Adrenal Medulla in stress response
The Role of the Adrenal Cortex in stress response
- Glucocorticoids
- Mineralocorticoids
Introduction.
The adrenal glands of vertebrates play a major role in the stress response, a state of threatened homeostasis.
Located on top of the kidneys (the renal organs), each adrenal gland is made up of two glands with different cell types, functions, and embryonic origins:
- the adrenal cortex, the outer portion,
- and the adrenal medulla, the central portion.
The adrenal cortex consists of true endocrine cells, whereas the secretory cells of the adrenal medulla develop from neural tissue.
Thus, like the pituitary gland, each adrenal gland is a fused endocrine and neuroendocrine gland.
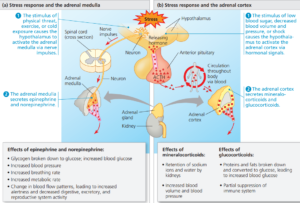
The Role of the Adrenal Medulla in stress response
Imagine that while walking in the woods at night you hear a growling noise nearby. “A Lion?” you wonder.
Your heart beats faster, your breathing quickens, your muscles tense, and your thoughts speed up.
These and other rapid responses to perceived danger comprise the “fight-or-flight” response.
This coordinated set of physiological changes is triggered by two hormones of the adrenal medulla,
- epinephrine (adrenaline)
- and norepinephrine (also known as noradrenaline).
Both are catecholamines, a class of amine hormones synthesized from the amino acid tyrosine.
Both molecules also function as neurotransmitters.
As hormones, epinephrine, and norepinephrine increase the amount of chemical energy available for immediate use.
Both catecholamines increase the rate of glycogen breakdown in the liver and skeletal muscles and promote the release of glucose by liver cells and fatty acids from fat cells.
The released glucose and fatty acids circulate in the blood and can be used by body cells as fuel.
Catecholamines also exert profound effects on the cardiovascular and respiratory systems.
For example;
They increase heart rate and stroke volume and dilate the bronchioles in the lungs, actions that raise the rate of oxygen delivery to body cells.
For this reason, doctors may prescribe epinephrine as a heart stimulant or to open the airways during an asthma attack.
Catecholamines also alter blood flow, causing constriction of some blood vessels and dilation of others.
The overall effect is to shunt blood away from the skin, digestive organs, and kidneys while increasing the blood supply to the heart, brain, and skeletal muscles.
How can epinephrine coordinate a response to stress that involves widely varying effects in individual tissues?
- In liver cells;
Epinephrine binds to a b-type receptor in the plasma membrane.
This receptor activates the enzyme protein kinase A, which in turn regulates enzymes of glycogen metabolism, causing the release of glucose into the blood.
- In the smooth muscle cells that line blood vessels supplying skeletal muscle;
The same kinase activated by the same epinephrine receptor inactivates a muscle-specific enzyme.
The result is smooth muscle relaxation, leading to vasodilation and hence increased blood flow to skeletal muscles.
- In the smooth muscle cells lining blood vessels of the intestines,
Epinephrine binds to an a-type receptor.
This receptor triggers a signalling pathway that involves enzymes other than protein kinase A and that causes smooth muscle contraction rather than relaxation.
The resulting vasoconstriction reduces blood flow to the intestines, facilitating the redirection of blood to active skeletal muscle.
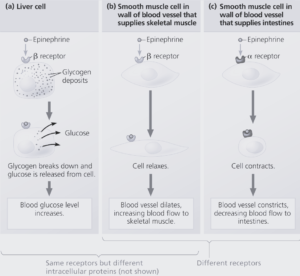
Thus, epinephrine elicits multiple responses if its target cells differ in the receptor protein they express or in the molecules activated by the receptor upon hormone binding.
The Role of the Adrenal Cortex in stress response
Like the adrenal medulla, the adrenal cortex mediates an endocrine stress response.
The two portions of the adrenal gland differ, however, in both the types of stress that trigger a response and the targets of the hormones that are released.
The adrenal cortex becomes active under stressful conditions that include low blood sugar, decreased blood volume and pressure, and shock.
Such stimuli cause the hypothalamus to secrete a releasing hormone that stimulates the anterior pituitary to release adrenocorticotropic hormone (ACTH), a tropic hormone.
When ACTH reaches the adrenal cortex through the bloodstream, it stimulates the endocrine cells to synthesize and secrete a family of steroids called corticosteroids.
The two main types of corticosteroids in humans are;
- glucocorticoids
- and mineralocorticoids.
Glucocorticoids
Glucocorticoids, such as cortisol, make more glucose available as fuel by promoting glucose synthesis from noncarbohydrate sources, such as proteins.
They also act on skeletal muscle, causing the breakdown of muscle proteins into amino acids.
These are transported to the liver and kidneys, converted to glucose, and released into the blood.
The synthesis of glucose upon the breakdown of muscle proteins provides circulating fuel when the body requires more glucose than the liver can mobilize from its glycogen stores.
If glucocorticoids are introduced into the body at a level above what is normally present, they suppress certain components of the body’s immune system.
For this reason, glucocorticoids are sometimes used to treat inflammatory diseases such as arthritis.
However, their long-term use can have serious side effects on metabolism.
Nonsteroidal anti-inflammatory drugs (NSAIDs), such as aspirin and ibuprofen, are therefore generally preferred for treating chronic inflammatory conditions.
Mineralocorticoids
Mineralocorticoids act principally in maintaining salt and water balance.
For example, the mineralocorticoid aldosterone functions in the ion and water homeostasis of the blood.
Like glucocorticoids, mineralocorticoids not only mediate stress responses but also participate in homeostatic regulation of metabolism.